� Also, some impurities may remain dissolved in the solution even after filtration. � Crystallization is a favored technique to purify substances like sugar that decompose or may get charred when heated to dryness. � Eventually, the crystals of pure copper sulfate separate out.ĭo you find crystallization and evaporation similar in any way? Actually, crystallization is better and has several advantages over evaporation. � Finally cover the solution and leave it undisturbed, thus allowing it to cool for a few hours. � Now heat the filtrate so that the water evaporates and a saturated solution is obtained. � Then filter it to remove the insoluble impurities and transfer the filtrate to a china dish. � First of all, take approximately 5 grams of impure copper sulfate and dissolve it in the minimum amount of water. Let us understand the process of crystallization by purifying an impure sample of copper sulfate. In fact, when a crystal is formed it tends to exclude the impurities which remain in the solution. It is one of the most commonly used techniques for the purification of inorganic and organic solids. Crystals are the purest form of a substance having a definite geometrical shape, and the process that separates a pure solid from a solution in the form of its crystals is known as crystallization. No, In fact, to obtain pure copper sulfate from an impure sample, we use another method of separation known as Crystallization. Can any of the methods which we have already learned be used to obtain pure copper sulfate from an impure sample? We have learned many different types of methods for the separation of different types of mixtures. � In addition to these applications, distillation is also used to separate the mixture of ether and toluene, and� the mixture of hexane and toluene. � Production of essential oils and herbal distillate is carried out by steam distillation. � Large-scale industrial distillation is widely used in petroleum refineries, petrochemical, and natural gas processing plants. � Distillation is employed in food processing to produce distilled beverages. � On the other hand, the water is left behind in the distillation flask.ĭo you know there are several important applications of distillation? Let us discuss a few important ones. 
This acetone can now be collected from the condenser outlet. � See the acetone vaporizing and then condensing in the condenser. � Then heat the mixture slowly keeping a close watch on the thermometer. � Now observe carefully how the apparatus is arranged. 
� Firstly, take the mixture in a distillation flask fitted with a thermometer. Let us have a look at the procedure of separation using the distillation method. Thus the separation of the mixture is achieved.Ĭan you separate a mixture of acetone and water? Yes�.it can be achieved by distillation. 
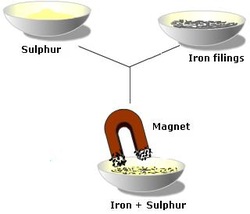
The more volatile component vapourises at its boiling point and then condenses in the condenser while the other component is left behind in the distillation flask. The separation of liquids is usually achieved at their respective boiling points. The process involves heating the liquid until boiling and then capturing and cooling the resultant vapors. Distillation is the process of separating miscible components of a mixture that do not decompose upon boiling and have sufficient differences in their boiling points. But do you know how a mixture of two miscible liquids is separated? Well�such a mixture can be separated by a method called Distillation. We know that different methods are used for the separation of different kinds of mixtures. In such cases, some special techniques are employed. However, sometimes the components of a mixture cannot be separated by simple methods. The components of a heterogeneous mixture can be separated by simple physical methods such as: A number of different methods are applied for separating the components of different kinds of mixtures. Is it possible to separate the constituent components of a mixture? Yes�it is. In fact, they are a mixture of two or more pure substances which are not chemically combined but are physically mixed in any random proportion. We already know that most natural substances are not chemically pure.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
